The trio sit in the same column within the transition metal hood.ģ. The Trio of Unreactive MetalsĪnd the least reactive metals we need to know are copper, silver, and gold. Lead in Group IV is also moderately reactive. Transition metals are generally less reactive than the main group metals. Within each group, the metal lower down the group is more reactive. They have the greatest tendency to lose electrons to form cations.įor the four metals we need to know, those from Group I are more reactive than those from Group II. Group I alkali metals and Group II alkaline earth metals are the most reactive. Ii) Reactions with acids: Reactive metals when react with acids produce the metal salt and hydrogen.The reactivity series is written into the Periodic Table! Highly Reactive Group I and II Metals Metals that don't react with water may react with acids. I) Reactions with cold water: The highly reactive metals easily react with cold water to form the metal hydroxide and hydrogen gas. The following reactions can be used to test the reactivity of metals Whereas, noble metals like silver, gold and platinum are nearly non-reactive. 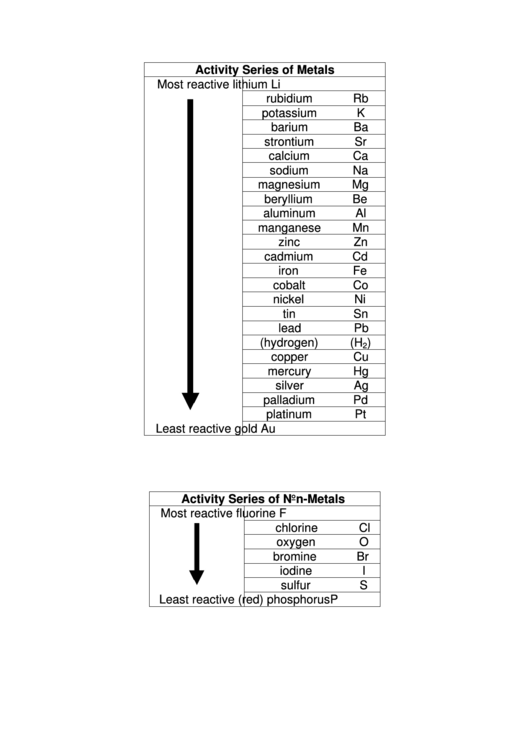
Generally, the alkali metals are the most reactive metals followed by the alkaline earth metals which are followed by transition metals. The reactivity series starts with the cesium metal which means cesium is at the top of the reactivity series or is the most reactive metal of this series. So, the reactivity series also tells the outcome of single displacement reactions, which are used to extract metals from their ores. Zn (s) + CuSO 4 (aq) → ZnSO 4 (aq) + Cu (s) For example, zinc being more reactive than copper displaces copper from copper sulphate as shown in the below reaction This is the reason for the displacement of less reactive metals by more reactive metals in the single displacement reactions. The more reactive metals or high ranking metals reduces (donate electrons) the ions of less reactive metals or low ranking metals in the series. iii) Displacement reaction between metals Similarly, we can predict the reaction of other metals located above lead with acids with the help of reactivity series. For example, when Zn is located above lead in the series when reacts with sulphuric acid leads to the formation of zinc sulfate and H 2 gas. Lead including the metals located above it, form salts when react with hydrochloric acid or sulphuric acid hydrogen gas is also produced. Similarly, we can predict the reaction of other metals with water with the help of reactivity series. For example, when potassium reacts with water potassium hydroxide and H 2 are produced as shown in the below chemical reaction For example, it can help you predict the outcome of reactions of metals with water, acids and with other metals as described below i) Reaction between metals and waterĬalcium including the metals that are situated above it in the series react with cold water to form the corresponding metal hydroxide and hydrogen gas. The reactivity series not only provides information about the properties and reactivity of metals but also offers various other applications. The metals that are located above the hydrogen can displace it from acids such as HCl and H 2SO 4. 
This is because hydrogen helps compare the reactivity of metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
